Ecological characteristics of symbiotic Symbiodiniaceae and bacteria in an environmentally sensitive reef-building coral Pocillopora sp.
-
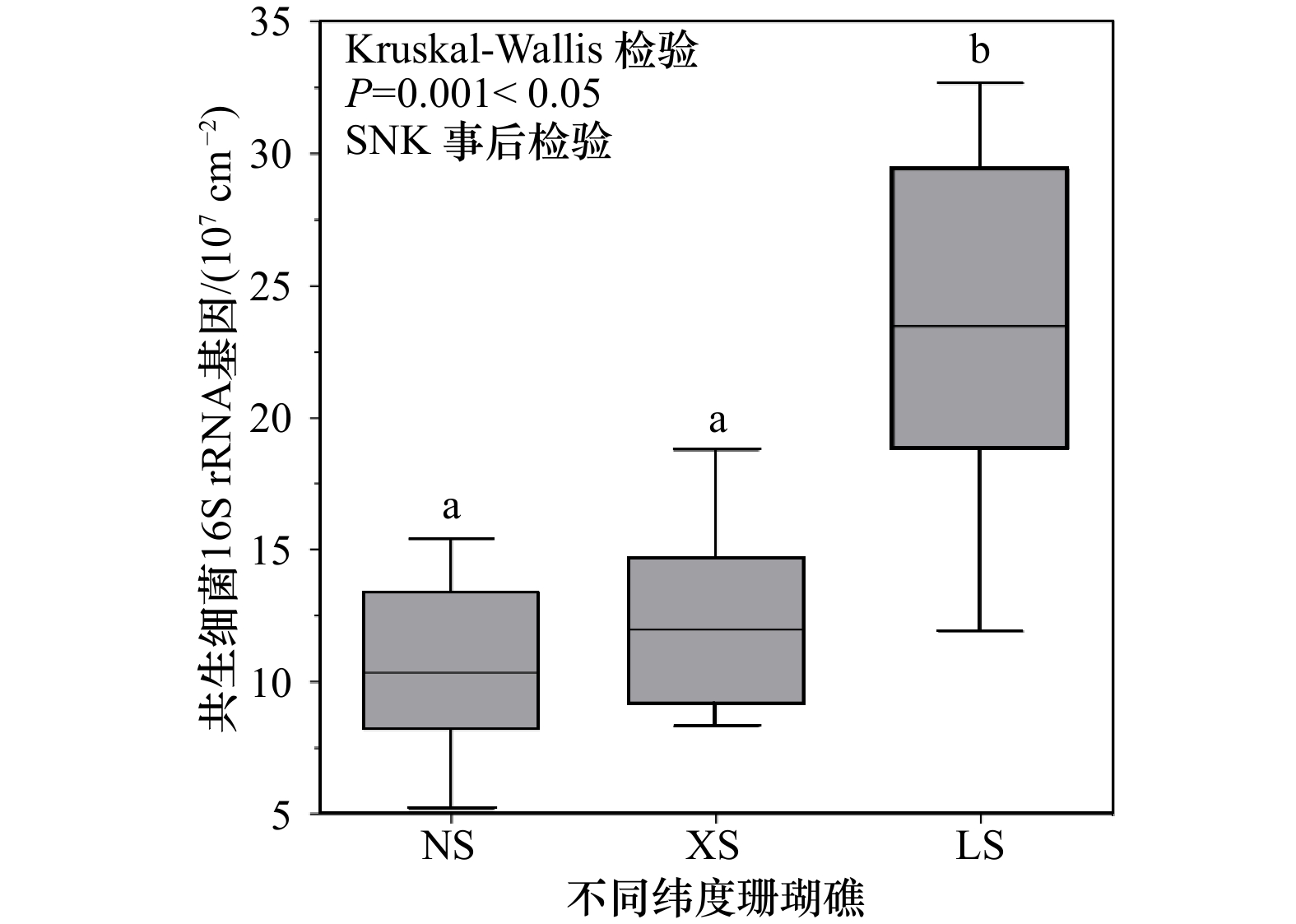
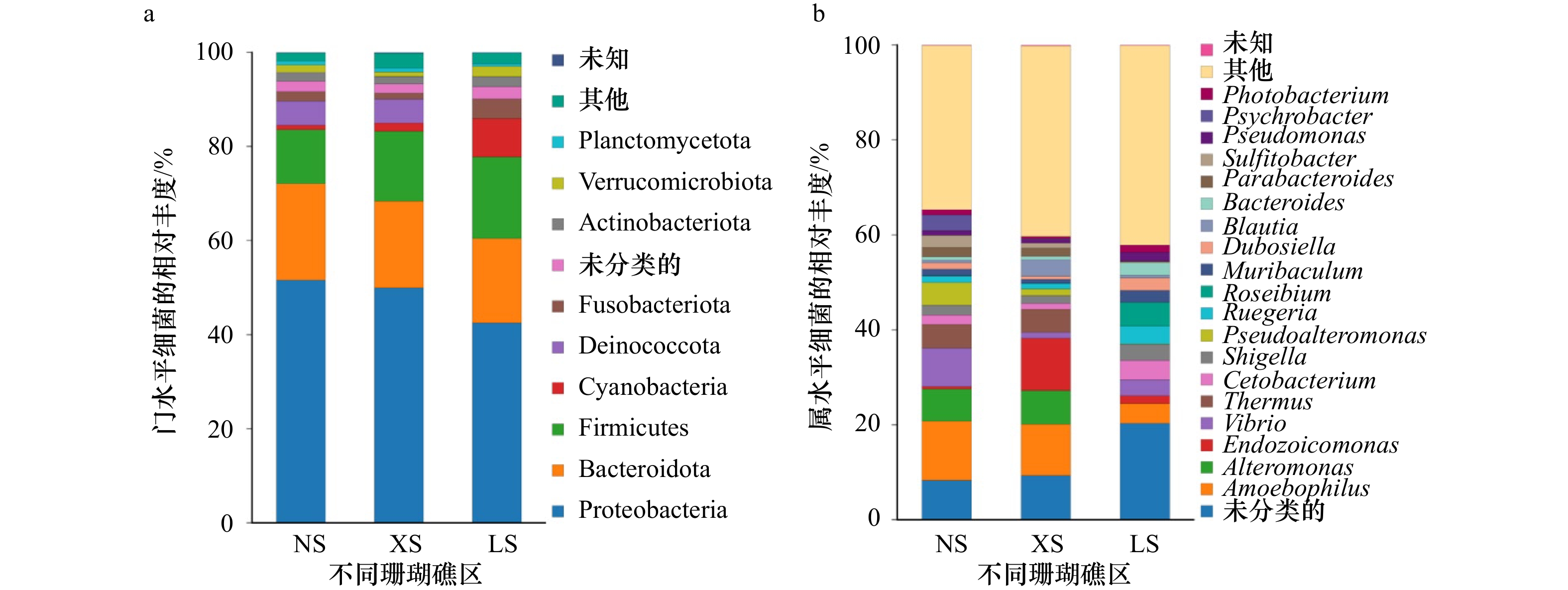
摘要: 在气候和人类活动的长期影响下,南海珊瑚礁演变出生境各异的珊瑚礁生态系统。珊瑚对特殊生境的适应离不开体内共生微生物类群的调节。分析共生微生物的生态特征有助于了解珊瑚的环境适应机制,同时可以对珊瑚未来的进化趋势做出评估。本研究从南海低纬度的南沙群岛(NS)和西沙群岛(XS)以及相对高纬度的海南陵水(LS)3个珊瑚礁区采集一种环境敏感型造礁珊瑚Pocillopora sp.,分析关键共生微生物(虫黄藻和细菌)的群落结构特征和生物量以及与环境的相关性。结果表明:(1)共生虫黄藻的群落组成在NS、XS与LS之间存在较大差异,NS和XS珊瑚共生虫黄藻的主导系群分别为D1、C1d和C42,而LS则为C42、C1d、C1p、C1和C1t;(2)共生细菌的群落组成均以变形菌(Proteobacteria)、拟杆菌(Bacteroidetes)和厚壁菌(Firmicutes)为主导系群,而具有光合作用功能的蓝细菌(Cyanobacteria)则随着纬度的升高,其相对丰度显著升高(从NS的0.95%到LS的8.18%,p<0.05);(3)在细胞数量方面,随着纬度的升高,共生虫黄藻的密度显著升高(从(3.39±0.49)×105 cells/cm2升至(8.90±0.65)×105 cells/cm2,p=0.001<0.05),在相对高纬度的LS同样有着更高的珊瑚共生细菌16S rRNA基因拷贝数(p=0.001<0.05);(4)环境因子相关性分析表明,具有潜在耐热性质的虫黄藻亚系群D1与海表温度和透明度呈正相关,而在LS珊瑚中占主导的虫黄藻C42以及蓝细菌的相对丰度则与海表温度呈显著负相关,与营养盐浓度呈显著正相关。本研究对南海的环境敏感型珊瑚Pocillopora sp.的环境适应微生态机制以及未来进化趋势提供了一个较全面的认识。Abstract: Under the long-term influence of climate change and human activities, coral reefs have evolved into special ecosystems with different habitats in the South China Sea. However, the adaptation of corals to special habitats is inseparable from the regulation of endosymbiotic microorganisms. The analysis of ecological characteristics of endosymbiotic microorganisms could help to understand the mechanism of coral host adaptation to environmental changes and the evolutionary trend. In this study, an environmentally sensitive reef-building coral Pocillopora sp. was collected from low latitude Nansha Islands (NS) and Xisha Islands (XS) and the relatively high latitude Hainan Lingshui (LS) in the South China Sea. The community composition and cells density, and environmental correlation of key endosymbiotic microorganisms (Symbiodiniaceae and bacteria) were analyzed. The results showed that: (1) there were great differences in the community composition of Symbiodiniaceae subclades among NS, XS, and LS. The dominant subclades in Pocillopora sp. from NS and XS were D1, C1d, and C42 respectively, while those from LS were C42, C1d, C1p, C1, and C1t; (2) the dominant bacterial compositions included Proteobacteria, Bacteroidetes, and Firmicutes in coral samples from different reef areas, while the relative abundance of Cyanobacteria with photosynthetic function increased significantly with the increase of latitude (from 0.95% of NS to 8.18% of LS, p<0.05); (3) in terms of the number of microbial cells in coral, the density of Symbiodiniaceae increased significantly with the increase of latitude (from (3.39±0.49)×105 cells/cm2 to (8.90±0.65)×105 cells/cm2 coral surface area, p=0.001<0.05), and there was also a higher absolute number of 16S rRNA gene copies in endosymbiotic bacteria in the relatively high latitude LS (p=0.001<0.05); (4) the correlation analysis of environmental factors showed that Symbiodiniaceae subclade D1 with potential thermal tolerance was positively correlated with sea surface temperature and transparency, while subclade C42 and Cyanobacteria were significantly negatively correlated with sea surface temperature and positively correlated with nutrient concentration. This study can provide insight into the micro-ecological mechanism of an environmentally sensitive coral Pocillopora sp. adaptation to environmental change and their evolutionary trend in the South China Sea.
-
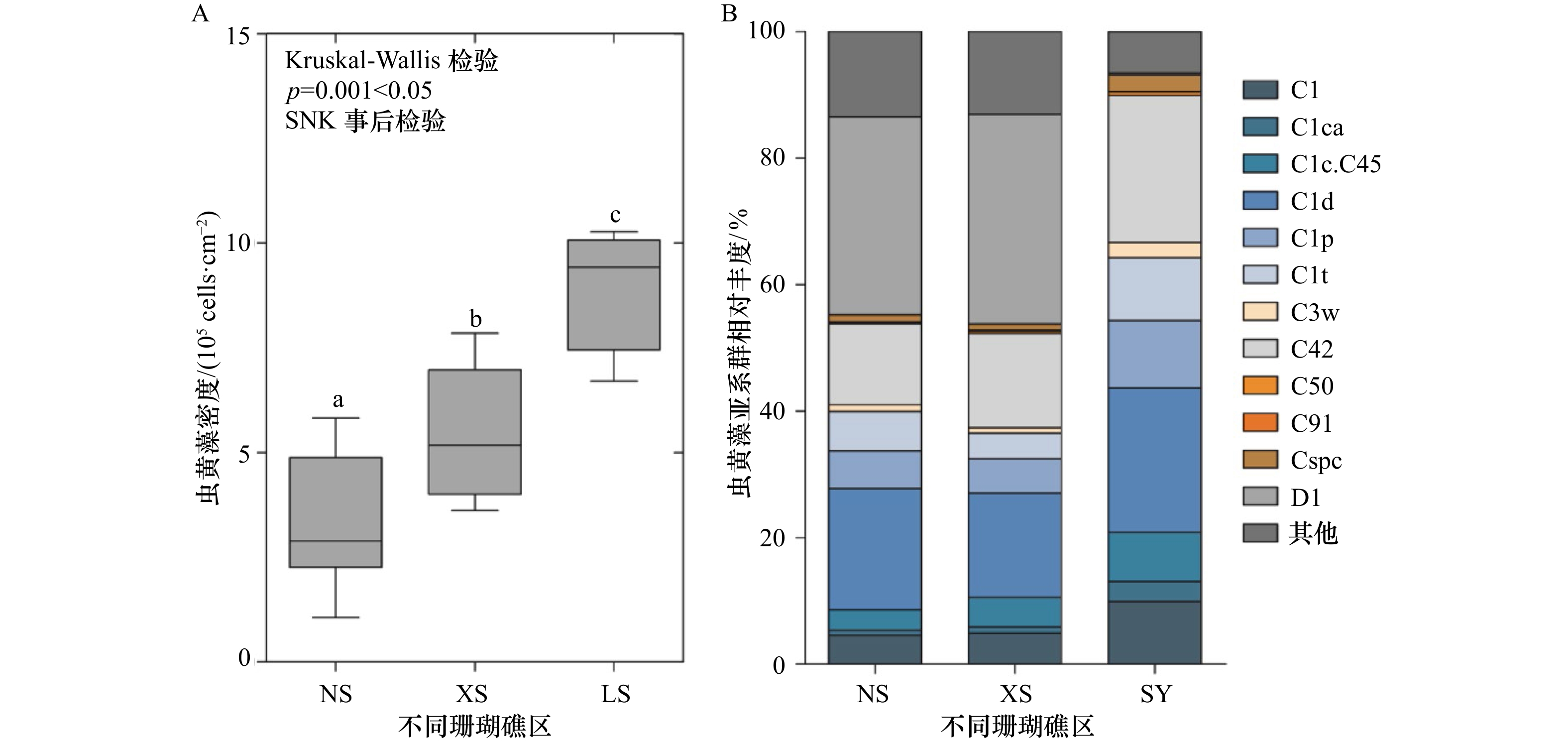
图 2 不同珊瑚礁区Pocillopora sp.共生虫黄藻密度(A)和基于ITS2序列分析的亚系群组成的变化(B)
从NS采集的样品数为20个,XS为19个,LS为11个;A中的数据为平均值±标准差;B中的“其他”汇总了相对丰度小于0.01%的所有虫黄藻亚系群成员;相同字母表示无显著差异;不同字母代表差异显著,p<0.05
Fig. 2 Change in Symbiodiniaceae density (A) and ITS2 sequence-based subclade composition (B) of Pocillopora sp. from different coral reef areas
The number of samples collected from NS, XS, and LS were 20, 19, and 11, respectively; the data in A were the means±SD; “others” in B represented Symbiodiniaceae subclades whose members had a relative abundance of less than 0.01%; same letters represent no significant difference; different letters represent significant difference, p<0.05
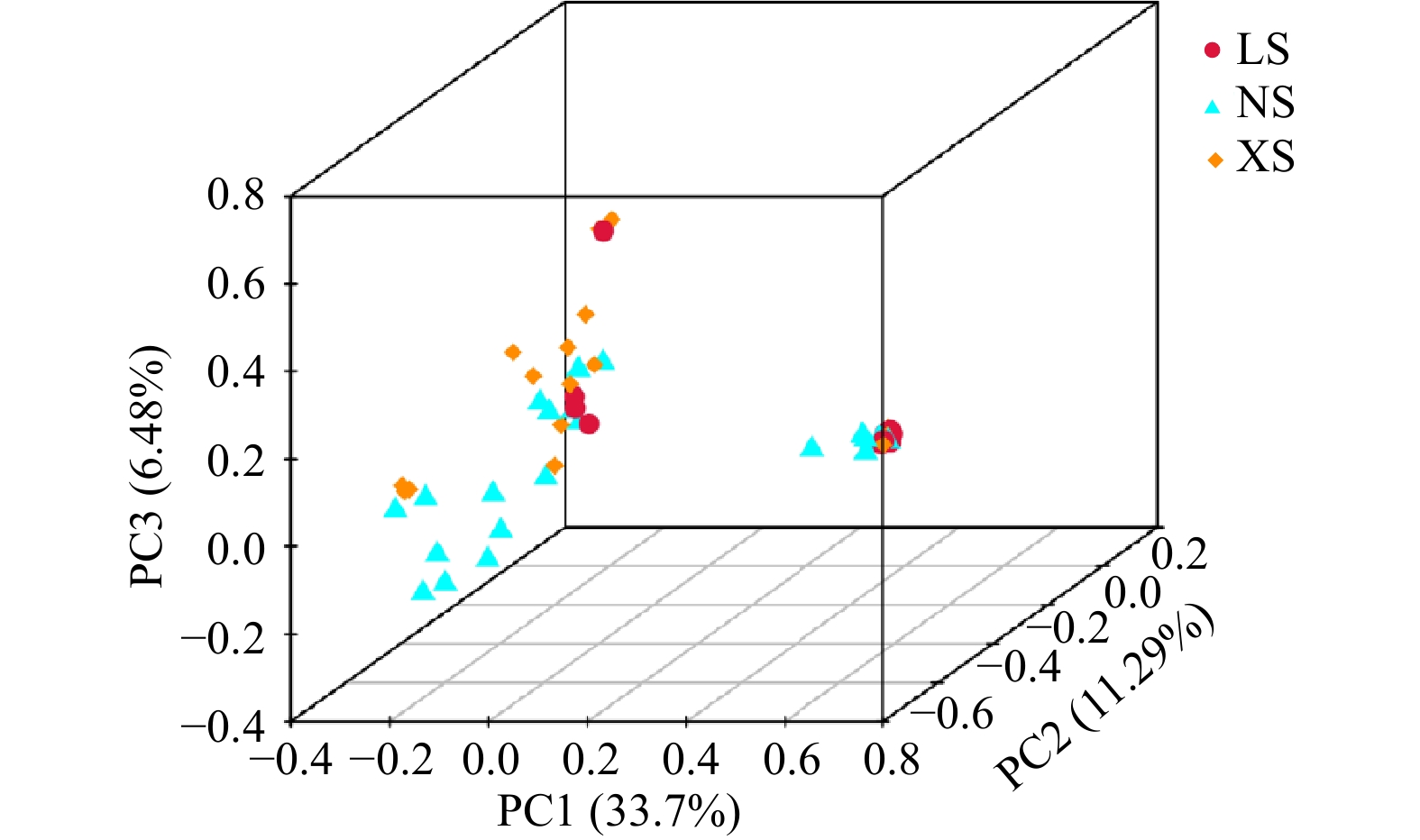
图 6 环境参数、珊瑚共生微生物生态参数和采样礁区之间的相关性
SRP:可溶性活性磷;PAR:光合有效辐射;DIN:溶解无机氮;POC:颗粒有机碳;Chl a:叶绿素a浓度;SST:海表温度
Fig. 6 Correlations among environmental parameters, ecological parameters of microorganisms, and sampling sites
SRP: Soluble reactive phosphorus; PAR: photosynthetic active radiation: DIN: dissolved inorganic nitrogen; POC: particulate organic carbon; Chl a: chlorophyll a concentration; SST: sea surface temperature
表 1 珊瑚共生细菌OTUs统计及Alpha多样性
Tab. 1 OTUs statistics and Alpha diversity of coral-associated bacteria
分组 OTU Shannon Simpson ACE Chao NS 825 5.02±0.38 0.87±0.03 225±30 213±30 XS 851 5.06±0.37 0.87±0.03 229±25 216±22 LS 570 5.35±0.70 0.87±0.05 275±51 268±53 注:相同珊瑚礁区样品的数据进行了合并分析;NS(n=20),XS(n=
19),LS(n=11)。 -
[1] Blackall L L, Wilson B, Van Oppen M J H. Coral-the world’s most diverse symbiotic ecosystem[J]. Molecular Ecology, 2015, 24(21): 5330−5347. doi: 10.1111/mec.13400 [2] Tsuchiya M, Galzin R, Davies N. Biodiversity research on coral reef and island ecosystems: scientific cooperation in the Pacific region[J]. Pacific Science, 2008, 62(3): 299−301. doi: 10.2984/1534-6188(2008)62[299:BROCRA]2.0.CO;2 [3] Wegley L, Edwards R, Rodriguez-Brito B, et al. Metagenomic analysis of the microbial community associated with the coral Porites astreoides[J]. Environmental Microbiology, 2007, 9(11): 2707−2719. doi: 10.1111/j.1462-2920.2007.01383.x [4] De Castro A P, Araújo S D Jr, Reis A M M, et al. Bacterial community associated with healthy and diseased reef coral Mussismilia hispida from Eastern Brazil[J]. Microbial Ecology, 2010, 59(4): 658−667. doi: 10.1007/s00248-010-9646-1 [5] Sweet M J, Croquer A, Bythell J C. Bacterial assemblages differ between compartments within the coral holobiont[J]. Coral Reefs, 2011, 30(1): 39−52. doi: 10.1007/s00338-010-0695-1 [6] Ceh J, Raina J B, Soo R M, et al. Coral-bacterial communities before and after a coral mass spawning event on Ningaloo Reef[J]. PLoS One, 2012, 7(5): e36920. doi: 10.1371/journal.pone.0036920 [7] Rosenberg E, Koren O, Reshef L, et al. The role of microorganisms in coral health, disease and evolution[J]. Nature Reviews Microbiology, 2007, 5(5): 355−362. doi: 10.1038/nrmicro1635 [8] Li Jie, Chen Qi, Long Lijuan, et al. Bacterial dynamics within the mucus, tissue and skeleton of the coral Porites lutea during different seasons[J]. Scientific Reports, 2014, 4: 7320. [9] Liang Jiayuan, Yu Kefu, Wang Yinghui, et al. Distinct bacterial communities associated with massive and branching scleractinian corals and potential linkages to coral susceptibility to thermal or cold stress[J]. Frontiers in Microbiology, 2017, 8: 979. doi: 10.3389/fmicb.2017.00979 [10] Qin Zhenjun, Yu Kefu, Wang Yinghui, et al. Spatial and intergeneric variation in physiological indicators of corals in the South China Sea: insights into their current state and their adaptability to environmental stress[J]. Journal of Geophysical Research:Oceans, 2019, 124(5): 3317−3332. doi: 10.1029/2018JC014648 [11] 左秀玲, 苏奋振, 王琦, 等. 全球变化下中国南海诸岛珊瑚礁热压力临时避难所研究[J]. 地理科学, 2020, 40(5): 814−822.Zuo Xiuling, Su Fenzhen, Wang Qi, et al. Thermal stress temporary refugia under global change for coral reefs in the South China Sea Islands[J]. Scientia Geographica Sinica, 2020, 40(5): 814−822. [12] Chen Biao, Yu Kefu, Liang Jiayuan, et al. Latitudinal variation in the molecular diversity and community composition of Symbiodiniaceae in coral from the South China Sea[J]. Front Microbiol, 2019, 10: 1278. doi: 10.3389/fmicb.2019.01278 [13] Chen Biao, Yu Kefu, Qin Zhengjun, et al. Dispersal, genetic variation, and symbiont interaction network of heat-tolerant endosymbiont Durusdinium trenchii: insights into the adaptive potential of coral to climate change[J]. Science of the Total Environment, 2020, 723: 138026. doi: 10.1016/j.scitotenv.2020.138026 [14] Liang Jiayuan, Yu Kefu, Wang Yinghui, et al. Diazotroph diversity associated with scleractinian corals and its relationships with environmental variables in the South China Sea[J]. Frontiers in Physiology, 2020, 11: 615. doi: 10.3389/fphys.2020.00615 [15] Chen Biao, Yu Kefu, Liao Zhiheng, et al. Microbiome community and complexity indicate environmental gradient acclimatisation and potential microbial interaction of endemic coral holobionts in the South China Sea[J]. Science of the Total Environment, 2021, 765: 142690. doi: 10.1016/j.scitotenv.2020.142690 [16] Tong Haoya, Cai Lin, Zhou Guowei, et al. Temperature shapes coral-algal symbiosis in the South China Sea[J]. Scientific Reports, 2017, 7: 40118. doi: 10.1038/srep40118 [17] Ziegler M, Seneca F O, Yum L K, et al. Bacterial community dynamics are linked to patterns of coral heat tolerance[J]. Nature Communications, 2017, 8: 14213. doi: 10.1038/ncomms14213 [18] Zhang Zhaojing, Qu Yuanyuan, Li Shuzhen, et al. Soil bacterial quantification approaches coupling with relative abundances reflecting the changes of taxa[J]. Scientific Reports, 2017, 7(1): 4837. doi: 10.1038/s41598-017-05260-w [19] Yu Kefu, Zhao Jianxin, Liu T S, et al. High-frequency winter cooling and reef coral mortality during the Holocene climatic optimum[J]. Earth and Planetary Science Letters, 2004, 224(1/2): 143−155. [20] Yu Kefu. Coral reefs in the South China Sea: their response to and records on past environmental changes[J]. Science China Earth Sciences, 2012, 55(8): 1217−1229. doi: 10.1007/s11430-012-4449-5 [21] Zhang Ruijie, Yu Kefu, Li An, et al. Antibiotics in corals of the South China Sea: occurrence, distribution, bioaccumulation, and considerable role of coral mucus[J]. Environmental Pollution, 2019, 250: 503−510. doi: 10.1016/j.envpol.2019.04.036 [22] Yu Kefu, Zhao Jianxin, Shi Qi, et al. Reconstruction of storm/tsunami records over the last 4 000 years using transported coral blocks and lagoon sediments in the southern South China Sea[J]. Quaternary International, 2009, 195(1/2): 128−137. [23] Yu Kefu, Zhao Jianxin, Collerson K D, et al. Storm cycles in the last millennium recorded in Yongshu Reef, southern South China Sea[J]. Palaeogeography, Palaeoclimatology, Palaeoecology, 2004, 210(1): 89−100. doi: 10.1016/j.palaeo.2004.04.002 [24] Li Jie, Long Lijuan, Zou Yiyang, et al. Microbial community and transcriptional responses to increased temperatures in coral Pocillopora damicornis holobiont[J]. Environmental Microbiology, 2021, 23(2): 826−843. doi: 10.1111/1462-2920.15168 [25] Zhou Guowei, Tong Haoya, Cai Lin, et al. Transgenerational effects on the coral Pocillopora damicornis microbiome under ocean acidification[J]. Microbial Ecology, 2021, 82(3): 572−580. doi: 10.1007/s00248-021-01690-2 [26] Zhao Meixia, Yu Kefu, Shi Qi, et al. The coral communities of Yongle atoll: status, threats and conservation significance for coral reefs in South China Sea[J]. Marine and Freshwater Research, 2016, 67(12): 1888−1896. doi: 10.1071/MF15110 [27] Qin Zhenjun, Yu Kefu, Liang Yangting, et al. Latitudinal variation in reef coral tissue thickness in the South China Sea: potential linkage with coral tolerance to environmental stress[J]. Science of The Total Environment, 2020, 711: 134610. doi: 10.1016/j.scitotenv.2019.134610 [28] Liao Zhiheng, Yu Kefu, Chen Biao, et al. Spatial distribution of benthic algae in the South China Sea: responses to gradually changing environmental factors and ecological impacts on coral communities[J]. Diversity and Distributions, 2021, 27(5): 929−943. doi: 10.1111/ddi.13243 [29] Guo Jing, Yu Kefu, Wang Yinghui, et al. Potential impacts of anthropogenic nutrient enrichment on coral reefs in the South China Sea: evidence from nutrient and chlorophyll a levels in seawater[J]. Environmental Science: Processes & Impacts, 2019, 21(10): 1745−1753. [30] 李淑, 余克服, 施祺, 等. 南海北部珊瑚共生虫黄藻密度的种间与空间差异及其对珊瑚礁白化的影响[J]. 科学通报, 2008, 53(2): 295−303. doi: 10.1007/s11434-007-0514-4Li Shu, Yu Kefu, Shi Qi, et al. Interspecies and spatial diversity in the symbiotic zooxanthellae density in corals from northern South China Sea and its relationship to coral reef bleaching[J]. Chinese Science Bulletin, 2008, 53(2): 295−303. doi: 10.1007/s11434-007-0514-4 [31] Lajeunesse T C, Trench R K. Biogeography of two species of Symbiodinium (Freudenthal) inhabiting the intertidal sea anemone Anthopleura elegantissima (Brandt)[J]. The Biological Bulletin, 2000, 199(2): 126−134. doi: 10.2307/1542872 [32] Bolger A M, Lohse M, Usadel B. Trimmomatic: a flexible trimmer for Illumina sequence data[J]. Bioinformatics, 2014, 30(15): 2114−2120. doi: 10.1093/bioinformatics/btu170 [33] Zhang Jiajie, Kobert K, Flouri T, et al. PEAR: a fast and accurate Illumina Paired-End reAd mergeR[J]. Bioinformatics, 2014, 30(5): 614−620. doi: 10.1093/bioinformatics/btt593 [34] Xu Lijia, Yu Kefu, Li Shu, et al. Interseasonal and interspecies diversities of Symbiodinium density and effective photochemical efficiency in five dominant reef coral species from Luhuitou fringing reef, northern South China Sea[J]. Coral Reefs, 2017, 36(2): 477−487. doi: 10.1007/s00338-016-1532-y [35] Edgar R C, Haas B J, Clemente J C, et al. UCHIME improves sensitivity and speed of chimera detection[J]. Bioinformatics, 2011, 27(16): 2194−2200. doi: 10.1093/bioinformatics/btr381 [36] Quast C, Pruesse E, Yilmaz P, et al. The SILVA ribosomal RNA gene database project: improved data processing and web-based tools[J]. Nucleic Acids Research, 2013, 41(D1): D590−D596. [37] Kõljalg U, Nilsson R H, Abarenkov K, et al. Towards a unified paradigm for sequence-based identification of fungi[J]. Molecular Ecology, 2013, 22(21): 5271−5277. doi: 10.1111/mec.12481 [38] Woo S, Yang Shanhua, Chen H J, et al. Geographical variations in bacterial communities associated with soft coral Scleronephthya gracillimum[J]. PLoS One, 2017, 12(8): e0183663. doi: 10.1371/journal.pone.0183663 [39] Gardner S G, Camp E F, Smith D J, et al. Coral microbiome diversity reflects mass coral bleaching susceptibility during the 2016 El Niño heat wave[J]. Ecology and Evolution, 2019, 9(3): 938−956. doi: 10.1002/ece3.4662 [40] Muscatine L, McCloskey L R, Marian R E. Estimating the daily contribution of carbon from zooxanthellae to coral animal respiration[J]. Limnology and Oceanography, 1981, 26(4): 601−611. doi: 10.4319/lo.1981.26.4.0601 [41] Falkowski P G, Dubinsky Z, Muscatine L, et al. Light and the bioenergetics of a symbiotic coral[J]. BioScience, 1984, 34(11): 705−709. doi: 10.2307/1309663 [42] Koren O, Rosenberg E. Bacteria associated with mucus and tissues of the coral Oculina patagonica in summer and winter[J]. Applied and Environmental Microbiology, 2006, 72(8): 5254−5259. doi: 10.1128/AEM.00554-06 [43] Ladrière O, Penin L, Van Lierde E, et al. Natural spatial variability of algal endosymbiont density in the coral Acropora globiceps: a small-scale approach along environmental gradients around Moorea (French Polynesia)[J]. Journal of the Marine Biological Association of the United Kingdom, 2014, 94(1): 65−74. doi: 10.1017/S0025315413001252 [44] Roth M S. The engine of the reef: photobiology of the coral-algal symbiosis[J]. Frontiers in Microbiology, 2014, 5: 422. [45] 张海洋, 赵美霞, 钟瑜, 等. 南海北部造礁石珊瑚共生体光合作用特征季节性监测[J]. 海洋地质前沿, 2021, 37(6): 84−91.Zhang Haiyang, Zhao Meixia, Zhong Yu, et al. Seasonal monitoring of photosynthetic characteristics of scleractinian corals in the Northern South China Sea[J]. Marine Geology Frontiers, 2021, 37(6): 84−91. [46] 许慎栋, 张志楠, 余克服, 等. 南海造礁珊瑚Favia palauensis营养方式的空间差异及其对环境适应性的影响[J]. 中国科学: 地球科学, 2021, 68(6): 839−852.Xu Shendong, Zhang Zhinan, Yu Kefu, et al. Spatial variations in the trophic status of Favia palauensis corals in the South China Sea: insights into their different adaptabilities under contrasting environmental conditions[J]. Science China Earth Sciences, 2021, 68(6): 839−852. [47] Kemp D W, Hernandez-Pech X, Iglesias-Prieto R, et al. Community dynamics and physiology of Symbiodinium spp. before, during, and after a coral bleaching event[J]. Limnology and Oceanography, 2014, 59(3): 788−797. doi: 10.4319/lo.2014.59.3.0788 [48] 董志军, 黄晖, 黄良民, 等. 两种造礁石珊瑚共生藻分子系统分类及其对水温升高的响应研究[J]. 海洋学报, 2009, 31(4): 141−148.Dong Zhijun, Huang Hui, Huang Liangmin, et al. Symbiotic algae in two scleractinian corals: molecular phylogeny and their response to elevated temperatures[J]. Haiyang Xuebao, 2009, 31(4): 141−148. [49] Ke Zhixin, Tan Yehui, Huang Liangmin, et al. Spatial distribution patterns of phytoplankton biomass and primary productivity in six coral atolls in the central South China Sea[J]. Coral Reefs, 2018, 37(3): 919−927. doi: 10.1007/s00338-018-1717-7 [50] Morrow K M, Moss A G, Chadwick N E, et al. Bacterial associates of two caribbean coral species reveal species-Specific distribution and geographic variability[J]. Applied and Environmental Microbiology, 2012, 78(18): 6438−6449. doi: 10.1128/AEM.01162-12 [51] Hoegh-Guldberg O. Climate change, coral bleaching and the future of the world’s coral reefs[J]. Marine and Freshwater Research, 1999, 50(8): 839−866. [52] Prabha R, Singh D P, Somvanshi P, et al. Functional profiling of cyanobacterial genomes and its role in ecological adaptations[J]. Genomics Data, 2016, 9: 89−94. doi: 10.1016/j.gdata.2016.06.005 [53] Harding K, Turk-Kubo K A, Sipler R E, et al. Symbiotic unicellular cyanobacteria fix nitrogen in the Arctic Ocean[J]. Proceedings of the National Academy of Sciences of the United States of America, 2018, 115(52): 13371−13375. doi: 10.1073/pnas.1813658115 [54] Santos H F, Carmo F L, Martirez N, et al. Cyanobacterial and microeukaryotic profiles of healthy, diseased, and dead Millepora alcicornis from the South Atlantic[J]. Diseases of Aquatic Organisms, 2016, 119(2): 163−172. doi: 10.3354/dao02972 -





 下载:
下载: